 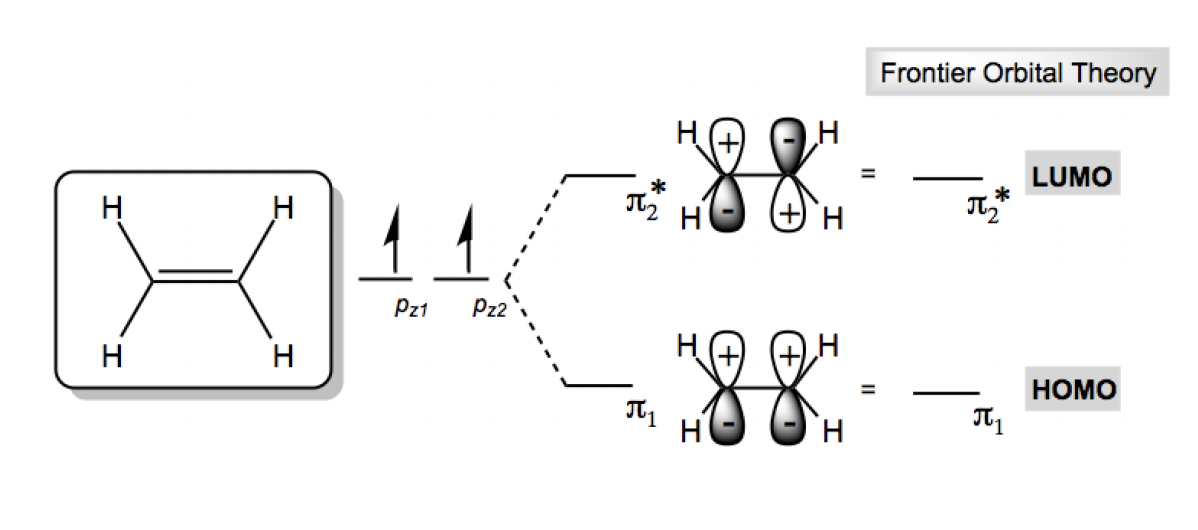
For example, in the case of a bromine molecule (Br2), the bond is broken by exposing it to light energy. By providing the energy equivalent of the energy gap from the outside, the electron moves into anti-bonding orbitals, resulting in an unstable state. It is often through overlap of the HOMO of. The energy difference between the HOMO and LUMO is called the HOMO-LUMO gap. Hence, smaller HOMO-LUMO gaps correspond to better stability. All molecules have a HOMO (highest occupied molecular orbital) and a LUMO (lowest unoccupied molecular orbital). The greater the mobility of the #pi# electrons in large conjugated pi orbital systems, the greater the distribution of the energy throughout the molecule, stabilizing it. When you have a large aromatic system in particular, small HOMO-LUMO gaps lead to mobile #\mathbf(pi)# electrons since it is easy for the electron to jump to a higher energy level that is close in energy. The larger the aromatic system is, the smaller the HOMO-LUMO gap! The same analogy exists between the LUMO level and the conduction band.The energy difference between the HOMO and LUMO level is regarded as band gap energy. Hence, it is the most important energy gap to consider.Įxcitations becomes easier as the HOMO-LUMO gap converges, such as for large aromatic systems (like tetracene or benzopyrene), or for transition-metal complexes (that is why they tend to be colored). The HOMO level is to organic semiconductors what the valence band is to inorganic semiconductors. In dieser Studie wird über die theoretische Analyse der Geometrien und elektronischen Eigenschaften verschiedener konjugierter Oligomere auf der Basis von Thiophen (Th) oder bicyclischen nichtklassischen Th-Einheiten berichtet. That means they are closest in energy out of all orbitals in the molecule.ĭue to the energies of these orbitals being the closest of any orbitals of different energy levels, the HOMO-LUMO gap is where the most likely excitations can occur. Die HOMO-LUMO-Lücke nimmt mit zunehmender Kettenlänge n in Oligomeren ab. Of the orbitals that have electrons, the HOMO is the highest in energy, and of the orbitals that don't, the LUMO is the lowest in energy. Don't be confused though, because the LUMO is higher in energy than the HOMO. HOMO stands for " Highest Occupied Molecular Orbital", and LUMO stands for " Lowest Unoccupied Molecular Orbital". 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
